Structure-fonction of nucleoproteic complexes of DNA topoisomerases
Structure-fonction of nucleoproteic complexes of DNA topoisomerases
We investigate the regulation of DNA topology by DNA topoisomerases that form large nucleoprotein complexes and catalyze relaxation of DNA entanglements in bacteria and human cells.
Our project is aiming at the functional and structural studies of DNA topoisomerase cellular complexes targeted by therapeutic compounds used in cancer and anti-bacterial therapies. The main goal of this study is to identify and study the structure/function of Topoisomerase-associated complexes targeted by drugs through a combination of functional and structural approaches.
Current projects
- Structural analysis of chromatin complexes of human DNA topoisomerases (TopoII).
- Impact of post-translational modifications on the catalytic activities of the human Topo II isoforms.
- Structure/function analysis of nucleoproteic complexes of the bacterial DNA gyrase. Check our latest article in Science 2024 : here
- Interplay of DNA topology and transcription (in collaboration with the team of Albert Weixlbaumer at IGBMC)
Awards and recognitions
Check our latest article published in Science, April 12th 2024 !
https://www.science.org/stoken/author-tokens/ST-1818/full
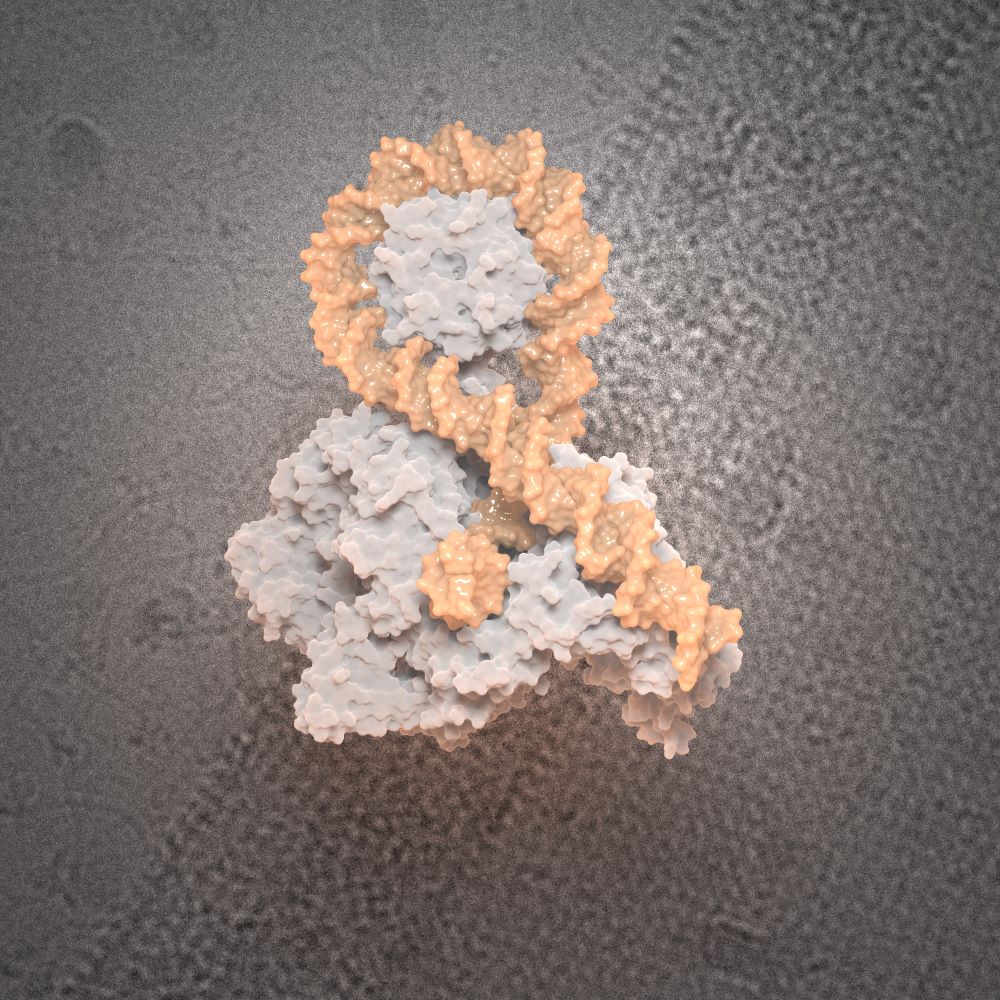
DNA gyrase captures a positive crossover prior to taking action.
Members
Researchers
PhD students
Publications
-
2024
-
DNA topoisomerase I acts as supercoiling sensor for transcription elongation in E. coli
- Vita Vidmar
- Céline Borde
- Lisa Bruno
- Maria Takacs
- Claire Batisse
- Charlotte Saint-André
- Chengjin Zhu
- Olivier Espéli
- Valérie Lamour
- Albert Weixlbaumer
BioRxiv
-
Structural basis of DNA crossover capture by Escherichia coli DNA gyrase
- Marlène Vayssières
- Nils Marechal
- Long Yun
- Brian Lopez Duran
- Naveen Kumar Murugasamy
- Jonathan Fogg
- Lynn Zechiedrich
- Marc Nadal
- Valérie Lamour
Science ; Volume: 384 ; Page: 227-232
-
-
2023
-
Inhibitors of UHRF1 base flipping activity showing cytotoxicity against cancer cells
- Stefano Ciaco
- Stefano Ciaco
- Viola Mazzoleni
- Aqib Javed
- Sylvia Eiler
- M. Ruff
- Marc Mousli
- Mattia Mori
- Yves Mély
Bioorganic Chemistry ; Volume: 137 ; Page: 106616
-
What’s on the Other Side of the Gate: A Structural Perspective on DNA Gate Opening of Type IA and IIA DNA Topoisomerases
- Vita Vidmar
- Marlène Vayssières
- Valérie Lamour
International Journal of Molecular Sciences ; Volume: 24 ; Page: 3986
-
-
2022
-
Thienoguanosine brightness in DNA duplexes is governed by the localization of itsππ* excitation in the lowest energy absorption band
- Stefano Ciaco
- Krishna Gavvala
- Vanille Greiner
- Viola Mazzoleni
- Pascal Didier
- Marc Ruff
- Lara Martinez-Fernandez
- Roberto Improta
- Yves Mély
Methods and Applications in Fluorescence ; Volume: 10
-
The human RNA polymerase I structure reveals an HMG-like docking domain specific to metazoans
- Julia L Daiß
- Michael Pilsl
- Kristina Straub
- Andrea Bleckmann
- Mona Höcherl
- Florian B Heiss
- Guillermo Abascal-Palacios
- Ewan P Ramsay
- Katarina Tlučková
- Jean-Clement Mars
- Torben Fürtges
- Astrid Bruckmann
- Till Rudack
- Carrie Bernecky
- Valérie Lamour
- Konstantin Panov
- Alessandro Vannini
- Tom Moss
- Christoph Engel
Life Science Alliance ; Volume: 5 ; Page: e202201568
-
-
2021
-
Structural basis for allosteric regulation of Human Topoisomerase IIα
- Arnaud Vanden Broeck
- Christophe Lotz
- Robert Drillien
- Léa Haas
- Claire Bedez
- Valérie Lamour
Nature Communications ; Volume: 12
-
Exonuclease VII repairs quinolone-induced damage by resolving DNA gyrase cleavage complexes
- Shar-Yin N. Huang
- Stephanie A. Michaels
- Brianna B. Mitchell
- Nadim Majdalani
- Arnaud Vanden Broeck
- Andres Canela
- Yuk-Ching Tse-Dinh
- Valérie Lamour
- Yves Pommier
Science Advances ; Volume: 7 ; Page: eabe0384
-
-
2020
-
Structural and Functional Studies of Human Type II Topoisomerases and their Post-Translational Modifications
- Christophe Lotz
- Claire Bedez
- Claire Batisse
- Arnaud Vanden Broeck
- Drillien Robert
- Marc Ruff
- Valerie Lamour
Biophysical Journal ; Volume: 118 ; Page: 532a
-
The interplay between DNA topoisomerase 2α post-translational modifications and drug resistance
- Christophe Lotz
- Valerie Lamour
Cancer Drug Resistance ; Page: 149-160
-

