RNA Diseases
RNA Diseases
It is estimated that up to 40 to 50% of the human genome is composed of repeated DNA elements, among which microsatellites are short repeated sequences of 3 to 6 nucleotides. Variations of DNA microsatellites size and sequence occur naturally and have important roles in genome evolution. However, variation in microsatellite size is also the source of devastating disorders, as more than 40 inherited developmental, neuromuscular and neurodegenerative human genetic diseases are caused by microsatellite expansions.
Studies in our group are focused on how microsatellite expansions located in the “non-coding” parts (5’UTR, Introns, 3’UTR, etc.) of the human genome cause genetic diseases.
Our team has a strong expertise and focus on the following neuromuscular and neurodegenerative pathologies:
Myotonic dystrophies (DM) type 1 and type 2 are the most common muscular dystrophy in adults. DM1 and DM2 are caused by large expansions of CTG or CCTG repeats located in DMPK 3’UTR or CNBP 1st intron, respectively. These CTG and CCTG repeats are transcribed into mutant CUG or CCUG RNA that bind and sequester the MBNL RNA binding proteins, resulting in specific RNA metabolism changes that cause the neuromuscular symptoms of these diseases. Publication of our group on myotonic dystrophies include Fugier et al., Nature Medicine, 2011; Rau et al., Nature Structural and Molecular Biology, 2011; Freyermuth et al., Nature Communication, 2016; Sellier et al., Nature Communication, 2018; etc.
Fragile X-Associated Tremor/Ataxia Syndrome (FXTAS) and Neuronal Intranuclear Inclusions Disease (NIID) are rare neurodegenerative disorders due to small expansions of GGC repeats located in the 5’UTR of the FMR1 and NOTCH2NLC genes, respectively. Importantly, we found that these GGC repeats are translated into novel polyglycine containing proteins, which are prone to form cellular inclusions that are toxic for neuronal cells. Publication of our group on these novel polyG diseases include Sellier et al., Cell reports, 2013 ; Sellier et al., Neuron, 2017 ; Boivin et al., Neuron, 2021; etc.
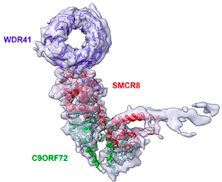
Amyotrophic Lateral Sclerosis and Frontotemporal Dementia (ALS-FTD) is the third neurodegenerative disease worldwide. The most common genetic cause of ALS-FTD is a large expansion of GGGGCC (G4C2) repeats in the first intron of the C9ORF72 gene. These G4C2 repeats are translated into proteins, which are highly toxic in cell and animal models. Furthermore, these G4C2 repeats promote DNA epigenetic changes that silence C9ORF72 expression. We found that the C9ORF72 protein belongs to a protein complex that regulates the activity of specific small GTPases, involved in vesicle trafficking and autophagy, a catabolic process essential for neuronal cells. Furthermore, we clarified the molecular mechanisms of G4C2 repeats translation into toxic proteins and found a novel synergic mechanism (2-hits) model of toxicity in ALS. Publication of our group on C9ORF72 and ALS include Sellier et al., EMBO Journal, 2016 ; Boivin et al., EMBO Journal, 2020 ; etc.
Our goals are to (1) Decipher the molecular causes of these diseases, (2) Establish relevant cellular (iPS, neurons, muscle cells, etc.) and animal (mouse) models of these diseases and (3) Identify drugs able to correct pathogenicity in these cell and animal models.
To achieve these goals, we developed a wide range of technique and strategies that include Molecular biology and Biochemistry approaches with protein purification and their functional analysis, Omics analyzes (transcriptomic and proteomic), Cell biology (primary human muscle cell culture, iPS from patients, primary mouse neuronal cultures, etc.) and analyzes of transgenic or knockout mouse models.
Hopefully, our work will help to clarify the causes and mechanisms of cell dysfunctions in these inherited disorders, but also provides novel cell and animal model to develop innovative therapeutic approaches for these devastating diseases.

Members
Researchers
PhD students
Engineers
Former members
- Charlotte FUGIER (Former Engineer)
- Angeline GAUCHEROT (Former Engineer)
- Frédérique RAU (Former PhD student)
- Fernande FREYERMUTH (Former PhD student)
- Michel NEY (Former PhD student)
- Camille CORBIER (Former PhD student)
Current projects
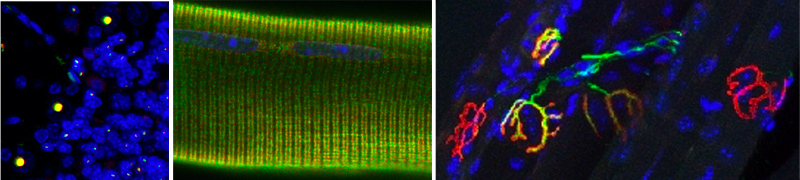
We have several new projects in the lab, either ongoing or in development
- We identified novel expanded repeats that are transcribed into mutant RNA in various related neurodegenerative diseases, and found that these repeats bind and sequester specific RNA binding proteins leading, in trans, to mRNA metabolism alterations that may explain the neuronal cell dysfunctions and the symptoms of these diseases. For reference works, this pathogenic mechanism is highly similar to titration of the MBNL RNA binding proteins by expansions of CUG RNA repeats resulting in specific RNA metabolism changes in myotonic dystrophies (Fugier et al., Nature Medicine 2011; Freyermuth et al., Nature Communication 2016; Sellier et al., Nature Communication 2018).
- We are studying novel GGC repeat expansions in rare neuromuscular and neurodegenerative diseases, and we are currently investigating whether these expansions are translated into novel and toxic polyglycine-containing proteins. For reference works, this is a direct extension of our work on FXTAS and NIID (Sellier et al., Neuron 2017; Boivin et al., Neuron 2021).
- Through extensive mass spectrometry analyzes, we identified novel proteins interacting with the C9ORF72 complex and we are investigating the functions and importance of these novel interactants for amyotrophic lateral sclerosis. We are also interested in solving the structure of this protein complex with these novel interactants. This work is a direct continuation of our previous studies on the C9ORF72 protein (Sellier et al., EMBO Journal 2016; Boivin et al., EMBO Journal 2020).
- Finally, we are investigating the molecular and cellular mechanisms of autophagic degradation of altered mitochondria (mitophagy), in relationship with neurodegenerative diseases.
Collaborations and networks
- Cecile MARTINAT (I-Stem, Paris), iPS and motor neurons cells models.
- Edor KABASHI (Imagine, Paris), Zebrafish animal models.
- Denis FURLING (Institut de Myologie, Paris), Pathogenic mechanisms in myotonic dystrophies.
- Patrick SCHULTZ (IGBMC, Strasbourg), CryoEM structure of the C9ORF72 protein complex.
Funding and partners

- 2021 - Foundation for Medical Research (FRM). Identification of a novel class of human genetic disease: the polyglycine (polyG) diseases.
- 2020 – Association pour la Recherche contre la Sclérose Latérale Amyotrophique (ARSLA). Molecular mechanism and therapeutic approach for a 2-hits model of toxicity in Amyotrophic Lateral Sclerosis (ALS).
- 2018 - French National Research Agency (ANR). Identification of novel mutations in Amyotrophic Lateral Sclerosis (ALS).
- 2017 - Thierry Latran Charity Foundation (TLF). Therapeutic approaches for C9ORF72-Amyotrophic Lateral Sclerosis (ALS).
- 2016 - French National Research Agency (ANR). Role of C9ORF72 in Amyotrophic Lateral Sclerosis (ALS).
- 2014 - French National Research Agency (ANR). Study of microRNA and mitochondrial alterations in Fragile X Tremor Ataxia Syndrome (FXTAS).
- 2012 - European Research Council (ERC) Starting grant. Understanding the RNA gain of function diseases.
- 2010 - French Foundation Against Myopathies (AFM). Mis-regulation of non-coding RNA in myotonic dystrophies.
- 2010 - European ERA-Net for Research on Rare Diseases (E-Rare). Understanding the causes of heart defects in myotonic dystrophies.
- 2008 - French Foundation Against Myopathies (AFM). Understanding the causes of muscular weakness in myotonic dystrophies.
- 2006 - INSERM AVENIR. Understanding the molecular causes of myotonic dystrophies.
News

Congratulations to David Pietri on the acceptance of his thesis!
Congratulations to David Pietri for the acceptance of his thesis! His work was directed by Nicolas Charlet-Berguerand and is titled: "Structure and…
Read more
Awards and recognitions

- 2018 - Award of scientific excellence. INSERM prime d'excellence scientifique.
- 2012 - European Research Council Starting grant.
- 2012 - INSERM DR2. Group leader permanent position.
- 2011 - Award of scientific excellence. INSERM prime d'excellence scientifique.
- 2006 - INSERM CR1. Scientist permanent position.
- 2006 - Award Georges Deflandre. Foundation de France.
- 2006 - INSERM AVENIR. Group starting position & funding.
Publications
-
2024
-
An unexpected polyglycine route to spinocerebellar ataxia
- Nicolas Charlet-Berguerand
Nature Genetics ; Volume: 56 ; Page: 1039-1041
-
-
2023
-
A biallelic loss of function variant in HORMAD1 within a large consanguineous Turkish family is associated with spermatogenic arrest
- Ozlem Okutman
- Manon Boivin
- Jean Muller
- Nicolas Charlet-Berguerand
- Stephane Viville
Human Reproduction ; Volume: 38 ; Page: 306-314
-
-
2022
-
PolyGA targets the ER stress-adaptive response by impairing GRP75 function at the MAM in C9ORF72-ALS/FTD
- Federica Pilotto
- Alexander Schmitz
- Niran Maharjan
- Rim Diab
- Adolfo Odriozola
- Priyanka Tripathi
- Alfred Yamoah
- Olivier Scheidegger
- Angelina Oestmann
- Cassandra Dennys
- Shrestha Sinha Ray
- Rochelle Rodrigo
- Stephen Kolb
- Eleonora Aronica
- Stefano Di Santo
- Hans Rudolf Widmer
- Nicolas Charlet-Berguerand
- Bhuvaneish Selvaraj
- Siddharthan Chandran
- Kathrin Meyer
- ...
Acta Neuropathologica ; Volume: 144 ; Page: 939-966
-
CGG repeat expansion in NOTCH2NLC causes mitochondrial dysfunction and progressive neurodegeneration in Drosophila model
- Jiaxi Yu
- Tongling Liufu
- Yilei Zheng
- Jin Xu
- Lingchao Meng
- Wei Zhang
- Yun Yuan
- Daojun Hong
- Nicolas Charlet-Berguerand
- Zhaoxia Wang
- Jianwen Deng
Proceedings of the National Academy of Sciences ; Volume: 119
-
De novo truncating NOVA2 variants affect alternative splicing and lead to heterogeneous neurodevelopmental phenotypes
- Marcello Scala
- Nathalie Drouot
- Suzanna C. Maclennan
- Marja W. Wessels
- Magdalena Krygier
- Lisa Pavinato
- Aida Telegrafi
- Stella A. de Man
- Marjon van Slegtenhorst
- Michele Iacomino
- Francesca Madia
- Paolo Scudieri
- Paolo Uva
- Thea Giacomini
- Giulia Nobile
- Maria Margherita Mancardi
- Ganna Balagura
- Giovanni Battista Galloni
- Alberto Verrotti
- Muhammad Umair
- ...
Human Mutation ; Volume: 43 ; Page: 1299-1313
-
Trinucleotide CGG Repeat Diseases: An Expanding Field of Polyglycine Proteins?
- Manon Boivin
- Nicolas Charlet-Berguerand
Frontiers in Genetics ; Volume: 13 ; Page: 843014
-
Evidence for a fragile X messenger ribonucleoprotein 1 ( FMR1 ) mRNA gain‐of‐function toxicity mechanism contributing to the pathogenesis of fragile X‐associated premature ovarian insufficiency
- Roseanne Rosario
- Hazel L. Stewart
- Nila Roy Choudhury
- Gracjan Michlewski
- Nicolas Charlet-Berguerand
- Richard A. Anderson
FASEB Journal ; Volume: 36 ; Page: e22612
-
-
2021
-
Translation of GGC repeat expansions into a toxic polyglycine protein in NIID defines a novel class of human genetic disorders: The polyG diseases
- Manon Boivin
- Jianwen Deng
- Véronique Pfister
- Erwan Grandgirard
- Mustapha Oulad-Abdelghani
- Bastien Morlet
- Frank Ruffenach
- Luc Negroni
- Pascale Koebel
- Hugues Jacob
- Fabrice Riet
- Anke Dijkstra
- Kathryn Mcfadden
- Wiley Clayton
- Daojun Hong
- Hiroaki Miyahara
- Yasushi Iwasaki
- Jun Sone
- Zhaoxia Wang
- Nicolas Charlet-Berguerand
Neuron ; Volume: 109 ; Page: 1825-1835.e5
-
Expression of expanded FMR1-CGG repeats alters mitochondrial miRNAs and modulates mitochondrial functions and cell death in cellular model of FXTAS
- Dhruv Gohel
- Lakshmi Sripada
- Paresh Prajapati
- Fatema Currim
- Milton Roy
- Kritarth Singh
- Anjali Shinde
- Minal Mane
- Darshan Kotadia
- Flora Tassone
- Nicolas Charlet-Berguerand
- Rajesh Singh
Free Radical Biology and Medicine ; Volume: 165 ; Page: 100-110
-
Neuropathology of FMR1 -premutation carriers presenting with dementia and neuropsychiatric symptoms
- Anke Dijkstra
- Saif Haify
- Niek Verwey
- Niels Prins
- Esmay van Der Toorn
- Annemieke Rozemuller
- Marianna Bugiani
- Wilfred den Dunnen
- Peter Todd
- Nicolas Charlet-Berguerand
- Rob Willemsen
- Renate Hukema
- Jeroen Hoozemans
Brain Communications ; Volume: 3 ; Page: fcab007
-

