Structure-fonction of nucleoproteic complexes of DNA topoisomerases
Structure-fonction of nucleoproteic complexes of DNA topoisomerases
We investigate the regulation of DNA topology by DNA topoisomerases that form large nucleoprotein complexes and catalyze relaxation of DNA entanglements in bacteria and human cells.
Our project is aiming at the functional and structural studies of DNA topoisomerase cellular complexes targeted by therapeutic compounds used in cancer and anti-bacterial therapies. The main goal of this study is to identify and study the structure/function of Topoisomerase-associated complexes targeted by drugs through a combination of functional and structural approaches.
Current projects
- Structural analysis of chromatin complexes of human DNA topoisomerases (TopoII).
- Impact of post-translational modifications on the catalytic activities of the human Topo II isoforms.
- Structure/function analysis of nucleoproteic complexes of the bacterial DNA gyrase. Check our latest article in Science 2024 : here
- Interplay of DNA topology and transcription (in collaboration with the team of Albert Weixlbaumer at IGBMC)
Awards and recognitions
Check our latest article published in Science, April 12th 2024 !
https://www.science.org/stoken/author-tokens/ST-1818/full
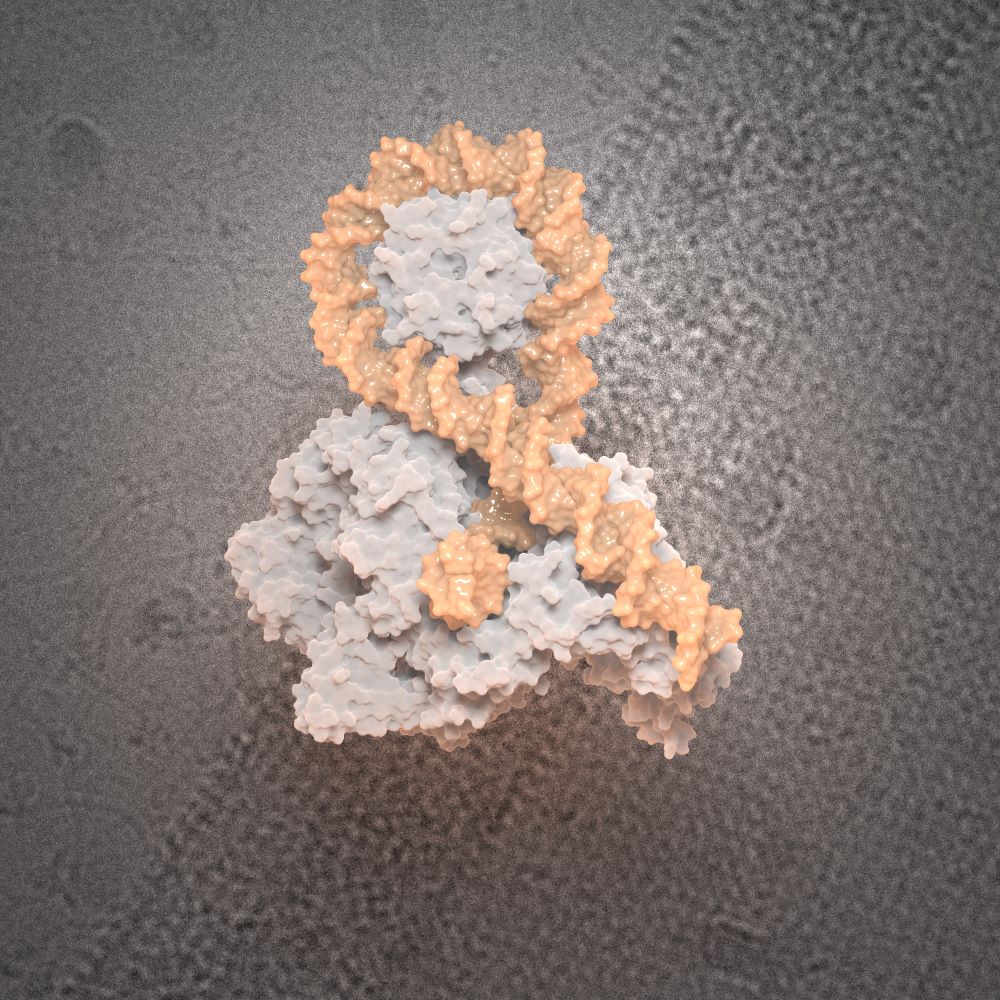
DNA gyrase captures a positive crossover prior to taking action.
Members
Researchers
PhD students
Publications
-
2010
-
Nondenaturing chemical proteomics for protein complex isolation and identification
- Ghyslain Budin
- Martin Moune-Dimala
- Geoffray Leriche
- Jean-Michel Saliou
- Julie Papillon
- Sarah Sanglier-Cianferani
- Alain van Dorsselaer
- Valérie Lamour
- Laurent Brino
- Alain Wagner
ChemBioChem ; Volume: 11 ; Page: 2359-2361
-
Computational tools in protein crystallography.
- Deepti Jain
- Valerie Lamour
Methods in Molecular Biology ; Volume: 673
-
-
2009
-
Crystal structure of the in vivo-assembled Bacillus subtilis Spx/RNA polymerase alpha subunit C-terminal domain complex.
- Valerie Lamour
- Lars F. Westblade
- Elizabeth A. Campbell
- Seth A. Darst
Journal of Structural Biology ; Volume: 168 ; Page: 352-6
-
-
2004
-
Proteolytic cleavage of the hyperthermophilic topoisomerase I from Thermotoga maritima does not impair its enzymatic properties
- Raynald Cossard
- Thierry Viard
- Valérie Lamour
- Michel Duguet
- Claire Bouthier de La Tour
BBA - Biochimica et Biophysica Acta ; Volume: 1700 ; Page: 161-170
-
Ultrahigh resolution drug design I: details of interactions in human aldose reductase-inhibitor complex at 0.66 A
- E Howard
- R Sanishvili
- Raul E. Cachau
- André Mitschler
- B Chevrier
- P Barth
- Valérie Lamour
- M van Zandt
- E Sibley
- C Bon
- Dino Moras
- Thomas R. Schneider
- Andrzejj Joachimiak
- Alberto Podjarny
Proteins - Structure, Function and Bioinformatics ; Volume: 55 ; Page: 792-804
-
-
2003
-
ATP-bound conformation of topoisomerase IV: a possible target for quinolones in Streptococcus pneumoniae
- Farid Sifaoui
- Valérie Lamour
- Emmanuelle Varon
- Dino Moras
- Laurent Gutmann
Journal of Bacteriology ; Volume: 185 ; Page: 6137-6146
-
-
2002
-
Crystallization of the 43 kDa ATPase domain of Thermus thermophilus gyrase B in complex with novobiocin
- Valérie Lamour
- L. Hoermann
- Jean-Marc Jeltsch
- Pierre Oudet
- Dino Moras
Acta Crystallographica Section D: Biological Crystallography ; Volume: 58 ; Page: 1376-1378
-
DNA gyrase interaction with coumarin-based inhibitors: the role of the hydroxybenzoate isopentenyl moiety and the 5'-methyl group of the noviose
- Daniel Lafitte
- Valérie Lamour
- Philippe O. Tsvetkov
- Alexander A. Makarov
- Michel Klich
- Pierre Deprez
- Dino Moras
- Claudette Briand
- Robert Gilli
Biochemistry ; Volume: 41 ; Page: 7217-7223
-
An open conformation of the Thermus thermophilus gyrase B ATP-binding domain
- Valérie Lamour
- Laurence Hoermann
- Jean-Marc Jeltsch
- Pierre Oudet
- Dino Moras
Journal of Biological Chemistry ; Volume: 277 ; Page: 18947-18953
-
-
2001
-
Hyperthermophilic topoisomerase I from Thermotoga maritima. A very efficient enzyme that functions independently of zinc binding
- Thierry Viard
- Valérie Lamour
- Michel Duguet
- Claire Bouthier de la Tour
Journal of Biological Chemistry ; Volume: 276 ; Page: 46495-46503
-
Page 3 of 4

