Differentiation and physiopathology of endocrine cells in the pancreas and intestine
Differentiation and physiopathology of endocrine cells in the pancreas and intestine
Our main goal is to decipher the transcriptional networks controlling the differentiation, function and maintenance of endocrine cells in the pancreas and intestine in normal and pathological conditions.
Future strategies for cell replacement therapies and regenerative medicine strongly depend on our knowledge of the detailed mechanisms that control the differentiation of multipotent stem cells into highly specialized cells. Along these lines of research, our goal is to understand how the diversity of pancreatic and intestinal endocrine cells is generated from stem/progenitor cells during embryogenesis towards adult life.
Pancreatic endocrine cells are clustered in islets embedded in the exocrine tissue. Islets contain five hormone producing cell-types, including insulin-secreting beta cells, which, in concert, control glucose homeostasis. Absolute or relative insulin-deficiency lead to diabetes. Intestinal endocrine cells, also called enteroendocrine cells (EECs), are very closely related to pancreatic islet cells with respect to their embryonic origin, differentiation programs and physiological roles in the control of energy homeostasis. EECs are rare cells found along the intestinal mucosa, they sense nutrients in the gut lumen and, in response, secrete a variety of hormones that act locally or at distance to regulate energy homeostasis via their control of intestinal absorption, food intake and insulin secretion. We showed previously that pancreatic and intestinal endocrine cells arise from progenitor cells expressing the pro-endocrine transcription factor Neurog3. In absence of Neurog3, islet cells and EECs do not form leading to neonatal diabetes and intestinal malabsorption in mice and human. Despite the identification of a few targets of Neurog3, the molecular mechanisms implementing Neurog3 endocrinogenic function are poorly understood.
We focus on the identification and study of novel effectors controlling endocrine sub-type specification and functional maturation. To tackle these questions, we use stem cell based human organoid culture systems and mouse models combined with gene editing and multi-omics approaches. We hope that our studies will contribute to the development of a cell-based therapy in diabetes, as well as to understand the mechanisms underlying the pathophysiology of islet and enteroendocrine hormone failures in human.
.png)
Members
Researchers
PhD students
Technicians
Funding and partners
The team was supported by founding from
- Novonordisk Foundation
- Agence National pour la Recherche (ANR)
- Fondation pour la Recherche Médicale (FRM)
- Novonordisk
- National Institute for Diabetes and Digestive and Kidney Disease (NIDDK)
- Société Française des Diabétiques (SFD)
- Juvenile Diabetes Research Foundation (JDRF)
- European Union (6th FP)
- Institut Benjamin Delessert
- Association Française des Diabétiques (AFD)
- Université de Strasbourg
- ARC
- INSERM-AVENIR
News
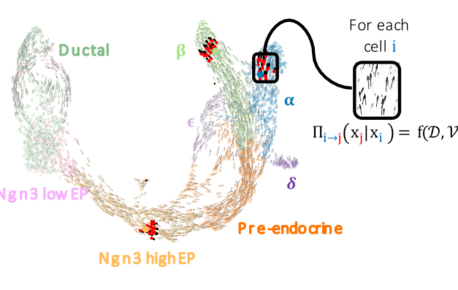
FateCompass: to identify and predict the decisive criteria of cell fate
Cell differentiation is a process regulated by gene expression through the action of transcription factors. Different transcription factors and…
Read more
Resources
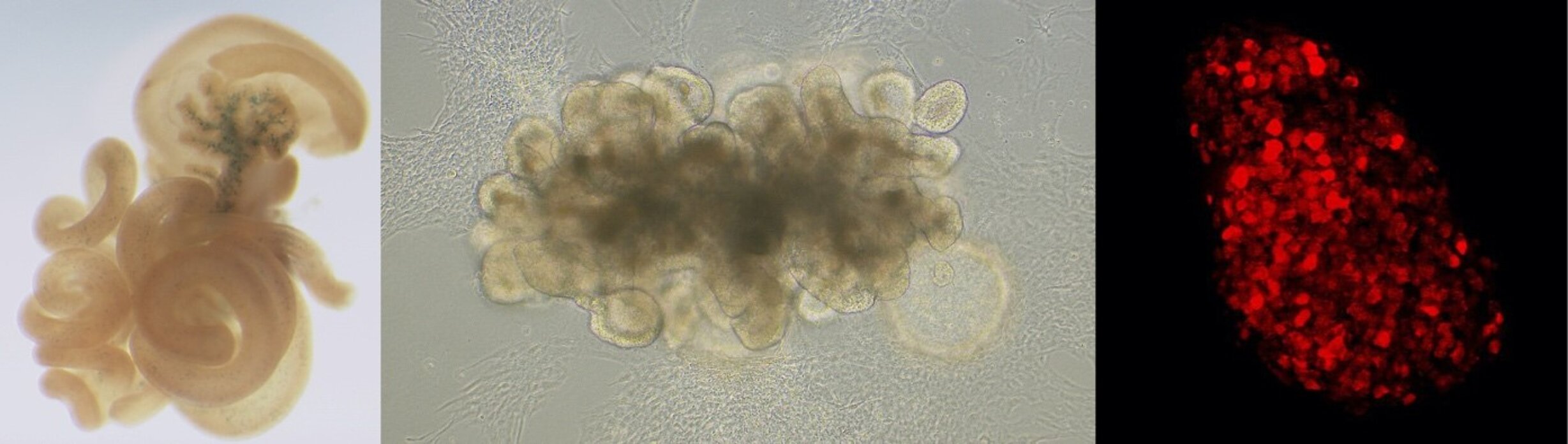
Human intestinal organoid. Enteroendocrine progenitors are expressing the transcription factor NEUROG3 (red).
https://seafile.igbmc.fr/f/80de9329f382470d80de/?dl=1
Publications
-
2003
-
Opposing actions of Arx and Pax4 in endocrine pancreas development
- Patrick Collombat
- Ahmed Mansouri
- Jacob Hecksher-Sorensen
- Palle Serup
- Jens Krull
- Gerard Gradwohl
- Peter Gruss
Genes and Development ; Volume: 17 ; Page: 2591-2603
-
Transcriptional program of the endocrine pancreas in mice and humans
- Klaus H. Kaestner
- Catherine S. Lee
- L. Marie Scearce
- John E. Brestelli
- Athanasios Arsenlis
- Phillip Phuc Le
- Kristen A. Lantz
- Jonathan Crabtree
- Angel Pizarro
- Joan Mazzarelli
- Deborah Pinney
- Steve Fischer
- Elisabetta Manduchi
- Christian J. Stoeckert
- Gerard Gradwohl
- Sandra W. Clifton
- Juliana R. Brown
- Hiroshi Inoue
- Corentin Cras-Méneur
- M. Alan Permutt
Diabetes ; Volume: 52 ; Page: 1604-1610
-
-
2002
-
Neurogenin3 is differentially required for endocrine cell fate specification in the intestinal and gastric epithelium
- Marjorie Jenny
- Céline Uhl
- Colette Roche
- Isabelle Duluc
- Valérie Guillermin
- Francois Guillemot
- Jan Jensen
- Michèle Kedinger
- Gerard Gradwohl
The EMBO Journal ; Volume: 21 ; Page: 6338-6347
-
-
2001
-
Crossregulation between Neurogenin2 and pathways specifying neuronal identity in the spinal cord
- Raffaella Scardigli
- Carol Schuurmans
- Gerard Gradwohl
- François Guillemot
Neuron ; Volume: 31 ; Page: 203-217
-
-
2000
-
Transcription Factor Hepatocyte Nuclear Factor 6 Regulates Pancreatic Endocrine Cell Differentiation and Controls Expression of the Proendocrine Gene <i>ngn3</i>
- Patrick Jacquemin
- Serge M. Durviaux
- Jan Jensen
- Catherine Godfraind
- Gerard Gradwohl
- François Guillemot
- Ole D. Madsen
- Peter Carmeliet
- Mieke Dewerchin
- Désiré Collen
- Guy G. Rousseau
- Frédéric P. Lemaigre
Molecular and Cellular Biology ; Volume: 20 ; Page: 4445-4454
-
neurogenin3 is required for the development of the four endocrine cell lineages of the pancreas
- Gérard Gradwohl
- Andrée Dierich
- Marianne Lemeur
- François Guillemot
Proceedings of the National Academy of Sciences of the United States of America ; Volume: 97 ; Page: 1607-1611
-
-
1999
-
Hes1 and Hes5 as notch effectors in mammalian neuronal differentiation
- Toshiyuki Ohtsuka
- Makoto Ishibashi
- Gerard Gradwohl
- Shigetada Nakanishi
- François Guillemot
- Ryoichiro Kageyama
EMBO Journal ; Volume: 18 ; Page: 2196-2207
-
-
1998
-
The bHLH Protein NEUROGENIN 2 Is a Determination Factor for Epibranchial Placode–Derived Sensory Neurons
- Carol Fode
- Gérard Gradwohl
- Xavier Morin
- Andrée Dierich
- Marianne Lemeur
- Christo Goridis
- François Guillemot
Neuron ; Volume: 20 ; Page: 483-494
-
-
1997
-
The activity of neurogenin1 is controlled by local cues in the zebrafish embryo
- P Blader
- Natacha Schroeder
- Gerard Gradwohl
- F Guillemot
- U Strähle
Development (Cambridge, England) ; Volume: 124 ; Page: 4557-4569
-
-
1996
-
Restricted Expression of a Novel Murineatonal-Related bHLH Protein in Undifferentiated Neural Precursors
- Gérard Gradwohl
- Carol Fode
- François Guillemot
Developmental Biology ; Volume: 180 ; Page: 227-241
-
Page 4 of 4

