Oncogenesis and progression of hormone-dependent cancers
Subgroup Leader : Daniel METZGER
Teams : Pathophysiological role of nuclear receptor signalling
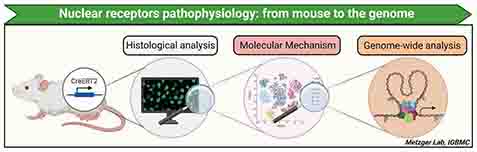
Steroid (e.g. androgens, estrogens, corticoids) and secosteroid (e.g. vitamin D) hormones play a key role in the development and cellular homeostasis of mammals. The activity of these hormones is mediated by proteins belonging to the nuclear receptor superfamily. The hormone-receptor pair functions as a highly selective lock-and-key system that modulates the expression of many genes and the activities of target cells.
Androgens and estrogens are essential for sexual maturation, corticoids are involved in the control of circadian activities and stress effects, and vitamin D regulates blood calcium levels. Moreover, due to their effects on metabolism, proliferation and inflammation, their receptors are therapeutic targets for pathologies refractory to current treatments such as various autoimmune and rare diseases, as well as cancers.
The objective of the laboratory is to understand the effects of these hormones under physiological and pathophysiological conditions in the whole organism. Our studies allow the characterization of the molecular and cellular events underlying the activity of these hormones, as well as the identification of new therapeutic targets for numerous pathologies with poor effective treatments.
Open post-doctoral position : www.igbmc.fr/igbmc/emplois/igbmc-post-doc-dmetzger-post-doctoral-position-at-the-institute-of-genetics-and-molecular-and-cellular-biology

PhD students (since 2010)
Post doctoral fellow (since 2010)
Engineer (since 2010)
General strategy
Nuclear receptors are transcription factors that transduce hormone signaling into a transcriptional response. The activity of about two-thirds of the 48 nuclear receptors is controlled by selective ligands (e.g. steroid hormones, retinoids, intracellular lipids), and is fine-tuned by transcriptional coregulators forming macromolecular complexes that activate (coactivators) or repress (corepressors) the expression of target genes by modulating the accessibility of the transcriptional machinery to DNA.
The various projects of the laboratory aim at understanding the effects of steroid hormones and vitamin D under physiological and pathophysiological conditions in the whole organism, using conditional somatic mutagenesis techniques (CreERT2) developed in the laboratory. Using an integrative biology approach combining phenotypic and genomic analyses, we characterize transcriptomic (e.g. RNA-seq) and proteomic (e.g. mass spectrometry) modifications, as well as the chromatin organization (e.g. ChIP-seq, ATAC-seq), in mouse models mimicking human pathologies. In parallel, we are developing single-cell analysis tools (e.g. flow cytometry, single-cell RNA-seq, ATAC-seq, spatial transcriptomics) to identify and characterize the cell populations involved in pathogenesis.
Benefits and impact
The various collaborations and our network of experts facilitate the validation of our results on human samples, and will improve our knowledge of the mechanisms underlying cellular homeostasis, as well as allow the development of new therapeutic options. In addition, a better understanding of the tissue-specific actions of these receptors will facilitate the design of new therapeutic agents with selective activities and thus fewer side effects.
Recent publication : doi.org/10.1038/s41419-025-07361-1
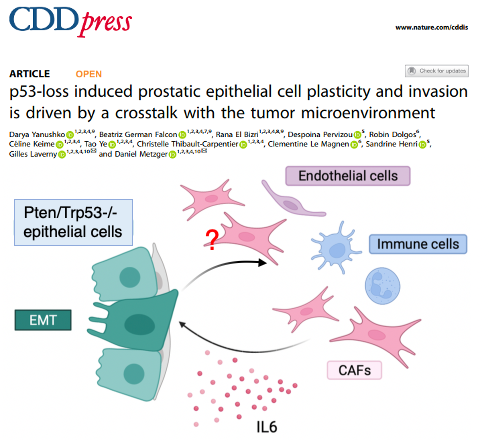
To visualize our single cell RNA seq data: https://metzger-chambon.igbmc.science/SingleCellViz/
Collaborations
Networks
![[Translate to English:] [Translate to English:]](/fileadmin/_processed_/c/d/csm_metzger2_82279dab4c.jpg)
On April 3, the research teams led by Drs Daniel Metzger and Gilles Laverny received a cheque for €10,000 to further their work on prostate cancer.
Read more
On
09/242025
https://zoom.us/j/99026487256?pwd=Y3ZkSFR5SVBzSExTajhRZVAveHEwZz09
Read more
Daniel Metzger
Post-doctorants
Doctorants
Nature Communications ; Volume: 11 ; Page: 6249
Antioxidants ; Volume: 8 ; Page: 168
Diabetes ; Volume: 68 ; Page: 1341-1352
Molecular & Cellular Oncology ; Volume: 6 ; Page: 1511205
Médecine/Sciences ; Volume: 34 ; Page: 904-906
Cell Metabolism ; Volume: 28 ; Page: 256-267.e5
Journal of Experimental Medicine ; Volume: 215 ; Page: 1749-1763
Frontiers in Physiology ; Volume: 9
Cell Metabolism ; Volume: 27 ; Page: 816-827.e4
Vitamin D, fourth edition: volume 1 – Biochemistry, Physiology and Diagnostics